Resources
Whitepaper
Turnkey Modular Manufacturing
With the recent downswing in the economy, as well as the current uncertainty about the affect of healthcare reform policy on the medical device and pharmaceutical industries, finding ways to improve efficiency and control costs has never been more important for manufacturers.
Enter your information below to download the whitepaper.

Whitepaper
What's your IQ OQ PQ validation IQ?
When it comes to packaging systems used for terminally sterilized medical devices, preserving sterility until use can literally be a matter of life and death. That’s why it’s so important for medical device manufacturers (MDMs) to ensure that the forming, sealing and assembly of sterile barrier systems (as well as the sterile barrier systems themselves) meet ISO 11607-2 requirements for IQ OQ PQ validation.
Enter your information below to download the whitepaper.

Sterile Barrier Solutions
TEQethylene™
Seeing the potential of HDPE as a viable solution for sterile barrier systems, TEQ worked with our suppliers to develop a better HDPE blend tray to combat these known issues. The result was a material with:
- Better clarity over conventional HDPE for improved product visibility and seal inspection
- Better processing and dimensional control
Enter your information below to download the whitepaper.
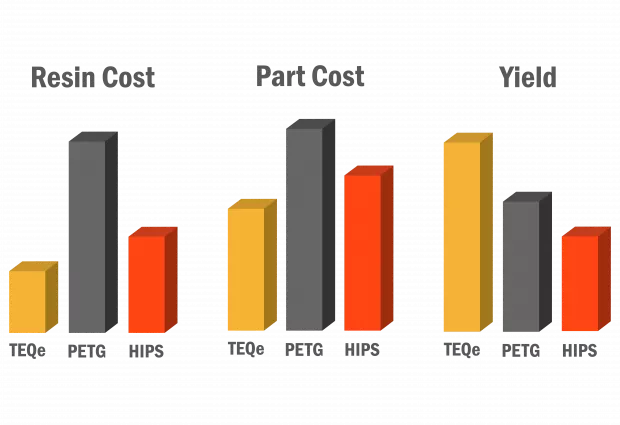
Sterile Barrier Solutions
TEQpropylene™
The next evolution of our TEQethylene™ Sterile Barrier System (SBS) Solution.
Enter your information below to download the whitepaper.

Sterile Barrier Solutions
TEQconnex™
Today’s medical packaging customers must meet a variety of unique and evolving demands from sterilization, regulatory requirements and sustainability - not to mention cost pressures. For this reason, it is essential for medical packaging manufacturers to continuously adapt their medical packaging and sterile packaging solutions to better meet these needs.
The evolution of TEQethylene™ to TEQconnex™ is an excellent example of such a transition.
Enter your information below to download the whitepaper.

Materials Guide
Our team has expertise in forming over 25 unique materials, including the latest biodegradable and recycled plastics.
Enter your information below to download our Materials Guide.

Haven't found what you're looking for?
Our expert team is here to help.






