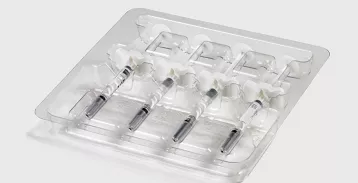
Your Global Partner in Precision
Our Process
TEQ works with every customer to create custom packaging and components that are the right material and meet their unique packaging demands, sustainability objectives and testing requirements. Explore our process, from design and development to tooling, production and logistics.

-
Sustainable Sterile Barrier Solutions
It’s essential for medical packaging manufacturers to continuously adapt their sterile packaging solutions to better meet the demands of regulatory requirements and sustainability. See how TEQ has evolved our materials in response to medical packaging challenges.
-
Multi-National Manufacturing Capabilities
We operate over 30 thermoforming lines across four facilities in the U.S. and Europe.
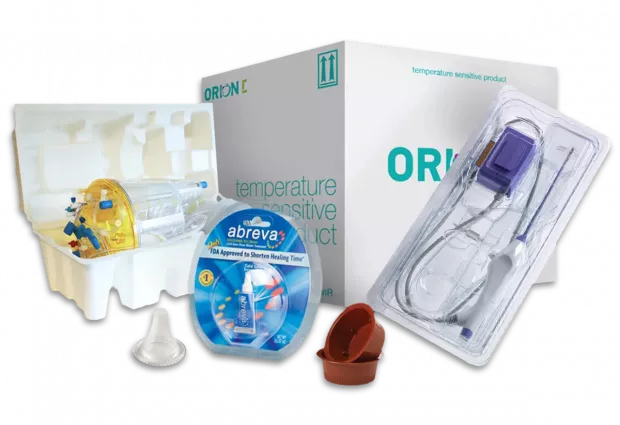
Sonoco TEQ Expands Total Solution Approach in Healthcare
Sonoco TEQ offers a range of cleanroom manufactured thermoformed and injection molded packaging and devices to the pharmaceutical and medical sectors.
From Our Partners
"Because of TEQ's extensive experience, in-house tooling capabilities, and superior knowledge, they can work with just about any product that can be thermoformed."
Markus Zlotosch, VP, Sales & Service, Kiefel Technologies
Thermoforming & Manufacturing Expertise
All Blog PostsHave a complex packaging challenge?
Find out how we can help.